Computational Electronics and Photonics Group
Integrated microstructures for label-free interrogation of protein conformational dynamics by plasmon-enhanced THz spectroscopy
Sensing and high throughput interrogation of protein functionality has become a key tool for state-of-the-art drug research and medical diagnostics. A variety of surface-based platforms have been developed that allow parallelized sensing assays. In addition to commonly used fluorescence detection, label-free techniques have become an attractive alternative for detection of biomolecular interactions. As example, refractometry based sensing in the visible and near infrared (IR) spectral region can quantify protein binding. At wavelengths in the IR and above the spectroscopic “finger-print” contains rich information on the protein structure and its constituents. A pressing question for the protein functionality is their conformational organization. Absorbance spectra at THz frequencies in the range of 0.1-2.0 THz show signatures from collective vibration modes of protein and hydrogen bonds, which are directly linked to the conformational structure of the protein. Current THz spectroscopy measurements however require substantial amounts of protein with several milligrams at millimolar concentrations, and therefore cannot unveil the mechanisms on a cellular level.
In an interdisciplinary collaboration, we aim to develop a sensor that overcomes these limitations, and ultimately allows label-free THz spectra from few protein molecules with high throughput and highest sensitivities. This is done by a plasmonic antenna with selectively deposited proteins [1] with nanometer precision in an aqueous environment. The antennas are fabricated using highly doped Germanium (Ge) on Silicon (Si) using CMOS compatible semiconductor processing technology [2].
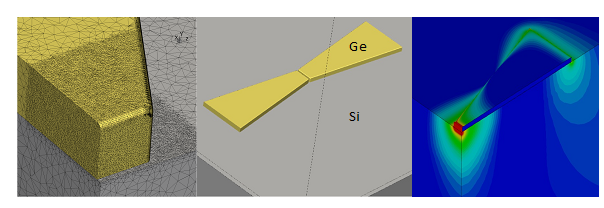
The contribution of the CEP group consists of a feasibility study and the design of the actual sensor based on theory and computational methods [3]. Starting from the accurate determination of the (complex) dielectric permittivity function for doped semiconductors at THz frequencies, the interaction of the loaded and unloaded sensor geometry with electromagnetic radiation is calculated. This is done with a fully three-dimensional realistic geometry of the structure, including features of the semiconductor process such as dopant variation, surface roughness and edge sharpness. Fig. 1 shows a plot of such a geometry.
A major result of the design study is shown in Fig. 2: with proper choice of material doping and sensor dimensions, the electromagnetic field intensity in a 1 mm gap of the antenna is enhanced by a factor of 8000, at a frequency of 0.5 THz, which enables strong focusing of the radiation on a subwavelength scale.
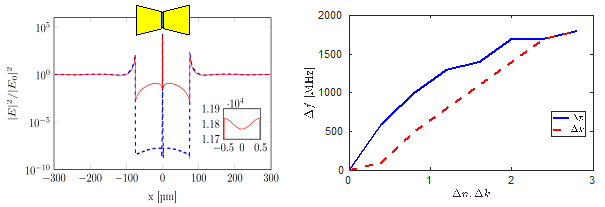
The right figure 2 shows the shift of the resonance frequency of the sensor (here: 0.5 THz) when depositing the sidewall of the antenna gap between the arms with bio material of 20 nm thickness. The horizontal axis is the change of real and imaginary part by the deposited material. As conclusion, highest sensitivity THz spectroscopy on sub-micrometer thick bio-films can be realized with the sensor concept. First experimental implementations show excellent agreement with the theoretical predictions for THz characterization.
Acknowledgements: This work has been performed in collaboration with IHP, Frankfurt, Oder and the Department of Biology/Chemistry, University of Osnabrück, and has been funded in part by the German Research Foundation (DFG) within the project ESSENCE SPP1857 (Electromagnetic Sensors for the Life Sciences).
References
- Kazmierczak, M., Flesch, J., Mitzloff, J., Capellini, G., Klesse, W. M., Skibitzki, O., You, C., Bettenhausen, M., Witzigmann, B., Piehler, J., Schroeder, T., and Guha, S., ' Stable and selective self-assembly of α- lipoic acid on Ge(001) for biomolecule immobilization', Journal of Applied Physics 123, 175305 (2018); doi: 10.1063/1.5022532
- M. Bettenhausen, F. Römer, B. Witzigmann, et al. (2018). Germanium Plasmon Enhanced Resonators for Label-Free Terahertz Protein Sensing. Frequenz, 72(3-4), pp. 113-122
- M. Bettenhausen, S. Grüßing, E. Hard, J. Flesch, F. Römer, C. Chavarin, W. Klesse, C. You, J. Piehler, G. Capellini, and B. Witzigmann, 'Impedance matching of Terahertz Plasmonic Antennas', International Journal of Infrared and Millimeter Waves doi.org/10.1007/s10762-019-00613-0
Prof. Dr. Bernd Witzigmann
full member

- Telephone
- +49 561 804-6543
- bernd.witzigmann[at]uni-kassel[dot]de
- Location
- Universität Kassel
Fachbereich 16 - Elektrotechnik & Informatik
Institut für Computational Electronics & Photonics
Wilhelmshöher Allee 71
34121 Kassel
- Room
- 2116
